What Is Used For Preservation Of Dissection Animals Now
By Richard E. Etheridge (adapted for the Www and updated by Grand. O'Brien and G. Schneider, May 1996)
Almost of the larger museums and universities that maintain preserved collections of reptiles and amphibians have curators trained in the approved methods of preparing and maintaining an alcoholic collection. On the other manus, many individuals with a non-professional interest in natural history have the inclination and opportunity to obtain and preserve herpetological specimens but lack knowledge of the proper techniques. It is the purpose of this article to be of help to these persons, for even pocket-sized collections casually assembled may be of great usefulness if the specimens are adequately labeled, well preserved, and fixed in a standard position.
Steps for the preservation of specimens for sclentific written report are as follows:
- Euthanizing. Specimens should be euthanized in a way that will leave them undamaged and relaxed, and follows best practices gear up forth past the Institutional Animal Intendance and Use Committee (IACUC).
- Injection and slitting. Liquid preservatives must exist introduced into the torso cavity, limbs and tail, either past hypodermic injection or through slits.
- Fixing. While the specimens are still relaxed, they should be arranged in trays so that they will harden in the proper position.
- Labeling. Each specimen should be accompanied by sure data, either attached directly or entered in a notebook with a number corresponding to a numbered tag tied to the specimen.
- Storage. Afterwards specimens take been fixed in the proper position, they should be stored in liquid preservative for at least several days, subsequently which they may be allowed to remain in the liquid, or transferred to plastic bags for temporary storage.
Preserving Solutions
Formalin: If at all possible, formalin should exist used for injecting and fixing specimens. Formalin is the commercial proper noun of a solution of formaldehyde gas (CH20) in water. Information technology is available at drugstores and chemic supply houses in the United states of america at a strength af from 38% to 40%. In Latin American countries, formalin may be purchased in many drugstores under the name "Formol" or "Formolina". Formalin must exist diluted with water before information technology is used every bit a preservative. A strength of ten% formalin is best for well-nigh purposes. If the original strength is 40%, it should be mixed at a ratio af nine parts h2o to ane part formalin. The advantages of formalin over other preservatives are: it is cheap, it is generally available, a small bulk af concentrated stock solution may be diluted as needed, and specimens most never decay in it. Its chief disadvantages are: it has a very irritating odor, it is very poisonous and may crusade pare irritation or rash, it has a tendency to brand specimens become brittle if the solution is besides strong, and tends to fade out sure colors speedily, and it must be stored in rustproof containers. (Buffering of the 10% solution is recommended as formalin is slightly acidic. I buffering system that may be used is a mixture of monobasic and dibasic Sodium Phosphate, at 13 gm/gallon [Monobasic] and 24 gm/gallon [Dibasic]).
Alcohol. At that place is a high federal tax on ethyl alcohol which makes it a very expensive preservative unless it can exist purchased past or through a university, museum or some other such establishment. Information technology is unremarkably sold at a force of 95% (190 proof). For injection and fixing it should be used at full strength. For storage of reptiles it should be used in the proportion of three parts 95% alcohol to one part h2o. Alcohol which has been stored in open containers loses its strength rapidly due to evaporation. Forcefulness may exist tested with an alcoholometer. Specimens which have been fixed in alcohol should exist carefully watched for signs of rotting. Alcoholic beverages, shaving lotions and Bay Rum incorporate ethyl alcohol. They should be used simply in an emergency and without dilution. Liquor which is 100 proof is only 50% ethyl booze.
Preparation: If specimens are to be made permanently allowed to decomposition, information technology is necessary that liquid preservative be introduced into the body crenel, limbs and tail inside as short a fourth dimension as possible after the animals take been killed. This may be accomplished either by injection (with a hypodermic syringe) or by making deep cuts with a sharp scalpel, razor blade or scissors. The most satisfactory mode is by injection. A x or twenty cc. syringe with a needle lock and several needles (guages 18 to 26) volition serve to inject most specimens.
Frogs and Toads: Injection should be made through the belly, directly into the body crenel. If the body is puffed with air, it should be deflated by gently squeezing with the fingers. Very small frogs require only a few drops of preservative; frogs 2 or 3 inches long just a few cc. Introduce only plenty preservative required to make the specimen look natural--it should not wait swollen. It is not necessary to inject the legs of any but the largest frogs. If equipment for injection is not available, a unmarried slit may be made in the belly, to ane side of the midline. The slit should be deep enough to allow free access of the preservative into the trunk cavity.
Salamanders. Virtually salamanders do not crave injection or slitting. If your specimens look "caved in" a small corporeality of preservative may exist injected into the trunk cavity, or a single slit fabricated in the abdomen to permit preservative.
Tadpoles. Tadpoles and small salamander larvae should always be preserved in ten% formalin, never in alcohol. But drop the tadpoles into formalin while they are still alive. Be sure there is enough preservative to encompass them and avoid overcrowding. After 24 hours all the liquid should be drained off and replaced with fresh formalin.
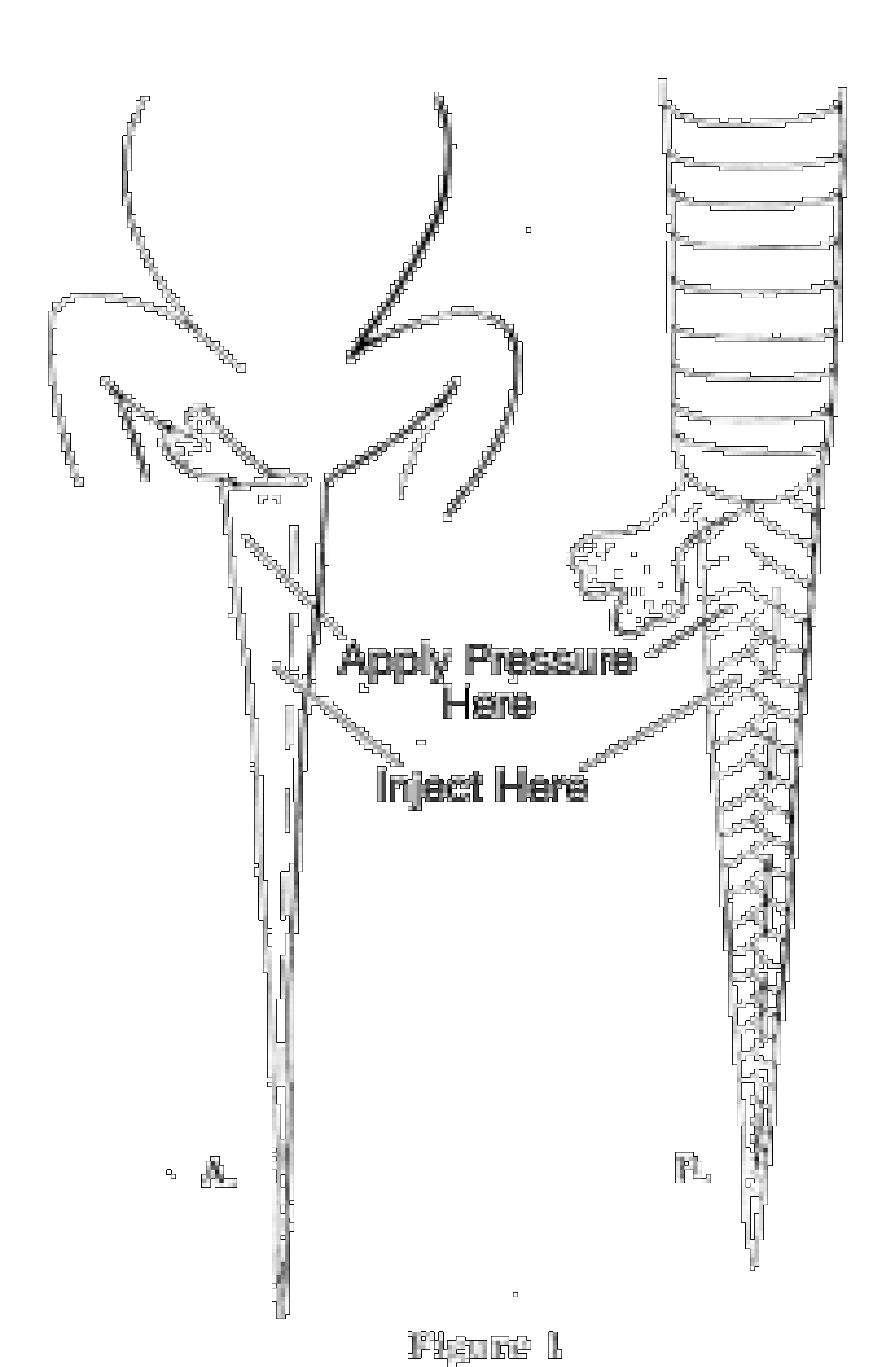 Lizards. Injection should be made through the belly direct into the body cavity. Intendance should be taken non to use besides much, or the body will become unnaturally distended. A series of slits should be fabricated in the under side of the tail with a sharp scalpel or razor blade. The slits should be from 1/8 to i/4 inch long and about 1/iv inch autonomously, and should extend from the base of the tail to the tip. Very big lizards must be injected or slit in the thicker portions of the limbs and neck. If space does not allow preservation of very large lizards whole, they may be skinned out, except for the head. To skin a big lizard, brand a cutting downward the abdomen from the neck to the base of the tail. Work the skin loose from the trunk, pulling the skin of the artillery and legs within out as far as the wrists and ankles. Do not attempt to peel out the caput, hands, feet or tail. Sever the wrists, ankles, neck and base of the tail, and remove the carcass. The skin should then he placed directly into preservative. If possible, one hemipenis of male lizards should be everted. This can exist accomplished hy injecting preservative into the base of the tail (before slitting) and at the same time applying pressure with the thumb only behind the anus (Fig. 1 A).
Lizards. Injection should be made through the belly direct into the body cavity. Intendance should be taken non to use besides much, or the body will become unnaturally distended. A series of slits should be fabricated in the under side of the tail with a sharp scalpel or razor blade. The slits should be from 1/8 to i/4 inch long and about 1/iv inch autonomously, and should extend from the base of the tail to the tip. Very big lizards must be injected or slit in the thicker portions of the limbs and neck. If space does not allow preservation of very large lizards whole, they may be skinned out, except for the head. To skin a big lizard, brand a cutting downward the abdomen from the neck to the base of the tail. Work the skin loose from the trunk, pulling the skin of the artillery and legs within out as far as the wrists and ankles. Do not attempt to peel out the caput, hands, feet or tail. Sever the wrists, ankles, neck and base of the tail, and remove the carcass. The skin should then he placed directly into preservative. If possible, one hemipenis of male lizards should be everted. This can exist accomplished hy injecting preservative into the base of the tail (before slitting) and at the same time applying pressure with the thumb only behind the anus (Fig. 1 A).
Snakes. Make a series of injections an inch or two apart through the belly into the trunk crenel. Begin merely behind the head and continue the injections to the anus. If a syringe is not available, a serial of slits must be made in the abdomen. For most snakes the slits should be about an inch apart and an inch long; smaller slits closer together for very pocket-sized snakes. Just as in lizards, a series of slits must be made in the under side of the tail and i hemipenis everted in males (Fig. 1B). Very large snakes may be skinned out, leaving the caput and tail attached. To skin a snake make a single, long cut in the abdomen, just to one side of the midline, showtime almost an inch behind the head and continuing to nearly an inch in front of the anus. Do not cut through the anal plate. Work the skin loose from the body, only do not endeavour to remove the pare from the caput or tail. Sever the body an inch behind the head and an inch in front of the anus, and (after recording the stomach contents, number of eggs, embryos, etc.) discard the carcass. Put a strip of cloth on the inner side of the peel and roll it upwards, beginning at the head. Tie the scroll with a piece of string and put it directly into preservative.
Alligators and Crocodiles. Pocket-sized individuals may exist preserved but as lizards. Larger individuals should be skinned out with the head fastened, rolled up and placed direct into preservative.
Turtles. Preservative should be injected into the body cavity just in front of each of the iv limbs, between the carapace and plastron. Use a long needle and continue injections until the head and · limbs are forced out of the vanquish. If a syringe is not bachelor, make deep cuts into the trunk cavity just in front of each leg. Limbs, neck and tail should be injected or slit, every bit in big lizards.
 Labels and Records: Specimens for which there are no information are of piffling or no scientific value. It is very important that each specimen be accompanied by sure information. This data may either be printed on a label which is attached to the specimen or may exist recorded in a notebook. If a notebook is used the data should be identified by a number; a tag bearing the aforementioned number should be attached to the specimen. The most important datum is the locality at which the collection was made. This should include the altitude from and direction to the center or city limits (land which) of the nearest city or town which can be easily institute on a map. Exercise not record distances to unincorporated towns or villages which are not likely to be marked on maps. If the altitude along a highway is used, state which highway. In the Usa, areas may be located with great accuracy past use of township, range and section maps. The name of the county and state, or of corresponding political units of foreign countries, should be included. Altitude may be of extreme importance and, if non readily ascertainable from maps of the surface area, information technology should be recorded. With the availability of inexpensive Global Positioning System (GPS) receivers, ane can now easily enter precise latitude and longitude data. This precise data is desired by those researchers engaged in mapping ranges of amphibians and reptiles with GIS mapping programs. Next in importance is the date of collection. The month should be written out or a clear abbreviation used.Practice not utilize numbers separated by dashes, such as "8-five-56". The proper name of the collector should be recorded. In addition to these data information technology is desirable to make careful descriptions of color and pattern from individuals before they are killed, since color often fades rapidly later on death.
Labels and Records: Specimens for which there are no information are of piffling or no scientific value. It is very important that each specimen be accompanied by sure information. This data may either be printed on a label which is attached to the specimen or may exist recorded in a notebook. If a notebook is used the data should be identified by a number; a tag bearing the aforementioned number should be attached to the specimen. The most important datum is the locality at which the collection was made. This should include the altitude from and direction to the center or city limits (land which) of the nearest city or town which can be easily institute on a map. Exercise not record distances to unincorporated towns or villages which are not likely to be marked on maps. If the altitude along a highway is used, state which highway. In the Usa, areas may be located with great accuracy past use of township, range and section maps. The name of the county and state, or of corresponding political units of foreign countries, should be included. Altitude may be of extreme importance and, if non readily ascertainable from maps of the surface area, information technology should be recorded. With the availability of inexpensive Global Positioning System (GPS) receivers, ane can now easily enter precise latitude and longitude data. This precise data is desired by those researchers engaged in mapping ranges of amphibians and reptiles with GIS mapping programs. Next in importance is the date of collection. The month should be written out or a clear abbreviation used.Practice not utilize numbers separated by dashes, such as "8-five-56". The proper name of the collector should be recorded. In addition to these data information technology is desirable to make careful descriptions of color and pattern from individuals before they are killed, since color often fades rapidly later on death.
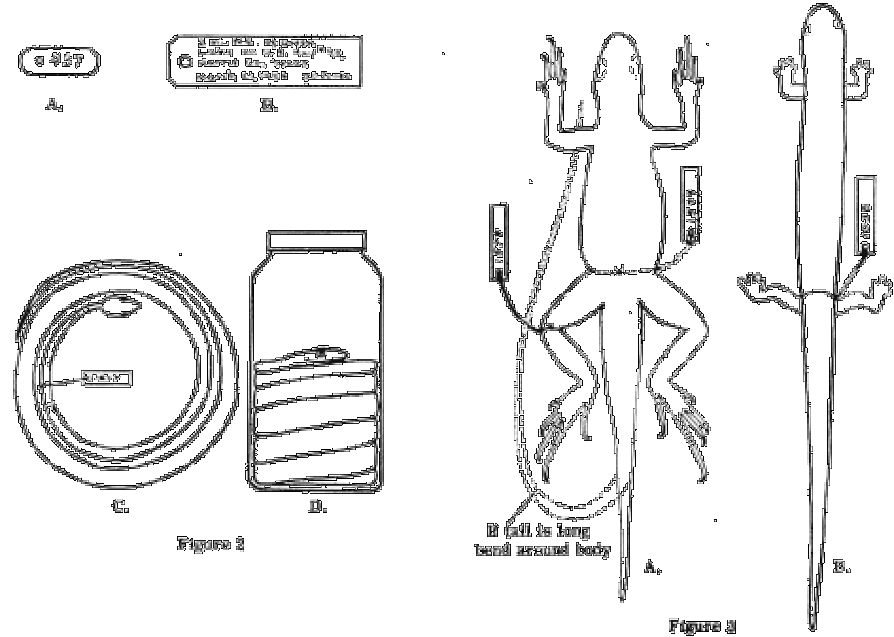
If a field notebook is used a description may be given of the habitat, climatic conditions and notes on beliefs, such as the vocalisation of calling frogs or toads, or a reference to an audiotaped telephone call. If numbered field tags are used, a notebook should be kept in waterproof ink or soft pencil, in which each number is listed consecutively, accompanied by the above information. Use tags that are especially fabricated for this purpose by biological supply houses, or utilise the best class of "bail" or linen paper. The size and shape recommended is shown inFig. 2 A. Tags fabricated of laminated paper or cardboard will autumn apart in liquid. Write only with a medium-soft pencil, never with ordinary ink, ball point pen or indelible pencil. "India Ink" or "Higgins Eternal Black" or Engrossing Ink may be used, but the tags should not be immersed in liquid until the ink is completely dry out. The new "Pigma pens" with blackness alcohol-proof ink seem to be a reasonable substitute for India Ink.
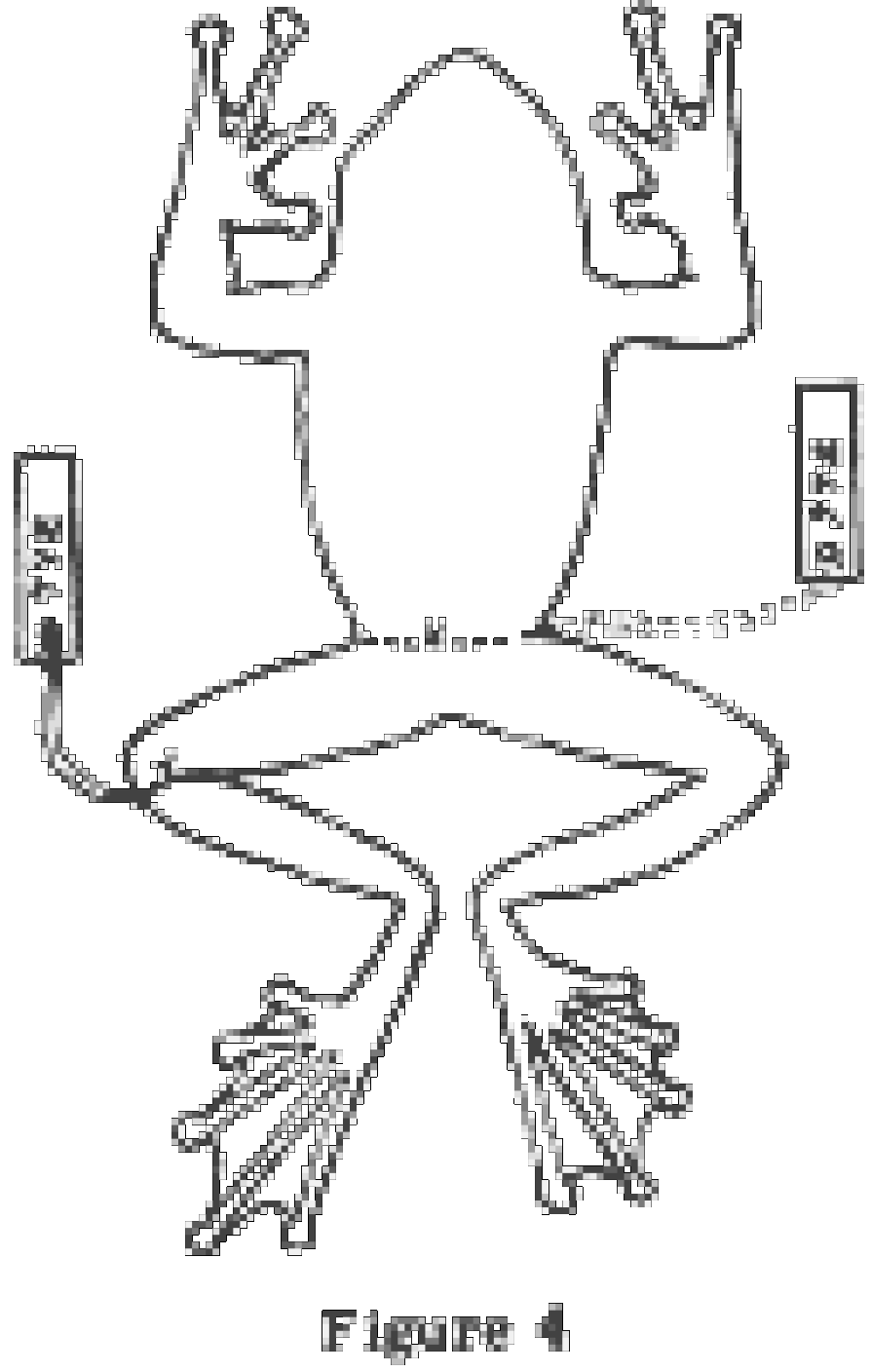
 If a notebook is non used, a tag large enough to bear the locality, date and collector's name should be attached to each specimen. The size and shape of such a tag is shown in Fig. 2 B. On lizards, frogs, toads and turtles, the tag should be tied (with a square knot) immediately beneath the genu of the left leg. Very small frogs, lizards and all salamanders should exist tied around the narrowest part of the waist. Snakes should exist tagged well back from the head but in forepart of the thickest part of the body. (Figs. two C, 3 A-B, 4 A). If a large series of specimens is collected at 1 locality on the same 24-hour interval, a unmarried tag may serve for the entire series. The tag should be tied on one of the specimens and the unabridged lot kept isolated from other specimens, either in a divide container or wrapped in fabric. This practise should be avoided if time permits individual tagging.
If a notebook is non used, a tag large enough to bear the locality, date and collector's name should be attached to each specimen. The size and shape of such a tag is shown in Fig. 2 B. On lizards, frogs, toads and turtles, the tag should be tied (with a square knot) immediately beneath the genu of the left leg. Very small frogs, lizards and all salamanders should exist tied around the narrowest part of the waist. Snakes should exist tagged well back from the head but in forepart of the thickest part of the body. (Figs. two C, 3 A-B, 4 A). If a large series of specimens is collected at 1 locality on the same 24-hour interval, a unmarried tag may serve for the entire series. The tag should be tied on one of the specimens and the unabridged lot kept isolated from other specimens, either in a divide container or wrapped in fabric. This practise should be avoided if time permits individual tagging.
 Reptiles and amphibians which accept been well-stock-still and immune to harden in the positions indicated in Figs. 2 C-D, 3 A-B and 4 A accept many advantages over those which have been left to harden in the posture in which they died. The extra time and care required is well worth the effort. Specimens can be more easily stored, photographed and examined and tin be more accurately measured and compared if they are allowed to harden in the proper position. The equipment required is not elaborate; several shallow trays of drinking glass, plastic or rust-proof metal, something with which to cover the trays, and newspaper. The bottom of the tray is covered with a single piece of paper (paper towels are good) which has been dampened with water. The specimens are placed on the newspaper, arranged in the desired position, and covered with another piece of damp newspaper. Pour enough preservative over the top to thoroughly soak the newspaper and permit about an eighth of an inch of liquid free in the bottom of the tray.
Reptiles and amphibians which accept been well-stock-still and immune to harden in the positions indicated in Figs. 2 C-D, 3 A-B and 4 A accept many advantages over those which have been left to harden in the posture in which they died. The extra time and care required is well worth the effort. Specimens can be more easily stored, photographed and examined and tin be more accurately measured and compared if they are allowed to harden in the proper position. The equipment required is not elaborate; several shallow trays of drinking glass, plastic or rust-proof metal, something with which to cover the trays, and newspaper. The bottom of the tray is covered with a single piece of paper (paper towels are good) which has been dampened with water. The specimens are placed on the newspaper, arranged in the desired position, and covered with another piece of damp newspaper. Pour enough preservative over the top to thoroughly soak the newspaper and permit about an eighth of an inch of liquid free in the bottom of the tray.
Specimens should exist injected (or slit) and tagged as soon as possible after they are expressionless and fixing should immediately follow. Individuals may be placed shut together on the tray but should not affect each other. The tray should be covered to prevent evaporation. Most amphibians will harden in a few hours, reptiles in ten or 12 hours. Big lizards, frogs and turtles may take a little longer, but the paper should be checked at least twice a day to be certain it is not drying out.
 Snakes. Pocket-size snakes may exist coiled flat in the tray if the coil does not exceed three and one half inches in its outside bore. The head should be within as in Fig. 2 C. Larger snakes should exist coiled in a jar and covered with preservative. If the snake has been injected it may be coiled with the belly down, tail at the bottom and head on top as in Fig. 2D. If slits are used, information technology should be coiled with the belly upwardly, caput on the lesser and tail on top. Tall, narrow bottles should be avoided; quart and pint sizes are best. Snakes also large to coil in a gallon jar should be skinned.
Snakes. Pocket-size snakes may exist coiled flat in the tray if the coil does not exceed three and one half inches in its outside bore. The head should be within as in Fig. 2 C. Larger snakes should exist coiled in a jar and covered with preservative. If the snake has been injected it may be coiled with the belly down, tail at the bottom and head on top as in Fig. 2D. If slits are used, information technology should be coiled with the belly upwardly, caput on the lesser and tail on top. Tall, narrow bottles should be avoided; quart and pint sizes are best. Snakes also large to coil in a gallon jar should be skinned.
Salamanders. Abdomen down, arms, legs and tail extended as in Fig. 3 B. A salamander tail volition oft twitch back and along long afterward the brute seems to be dead. X or xv minutes afterward they accept been laid out check to be sure the tail is still direct. Large specimens, 10 or more than inches in length, may exist coiled like snakes.
Frogs and Toads. Place the frog belly down, arms and legs extended as in Fig. four A. The fingers and toes should be separated and extended, especially if they are webbed. The inner margin of the forelimb from the elbow to the tip of the 4th finger should form a direct line. The sole of the foot may be up or downwards, whichever seems most natural (downwards in treefrogs and up in most other frogs and toads).
 Storage: Subsequently the specimens have been injected or slit, tagged, and fixed, they should be put direct into preservative. If they are to exist transferred later to plastic bags for temporary storage or to exist shipped they should first be allowed to remain completely immersed in preservative for at least 48 hours if formalin is used, or a calendar week if booze is used. The longer they are allowed to stay in preservative, the better. They should be loose and completely covered with plenty of liquid. Specimens which have been hardened in trays should as well exist allowed to soak in preservative for a day or 2 before being shipped or placed in plastic bags for storage. If space is no problem, preserved specimens are best kept in glass containers. Bond-top jars with a glass height and rubber gasket are best. Fruit jars with a metallic screwtop lid may be used only should be carefully watched for rust and evaporation. Drinking glass jars with polyethylene lids and liners are more normally used in collections, since the lids form a tight seal and are easier to obtain than the traditional bail-elevation jars. Metal containers should exist used simply for temporary storage unless coated on the inside with methane series, "Bakvar", or some other rustproof material.
Storage: Subsequently the specimens have been injected or slit, tagged, and fixed, they should be put direct into preservative. If they are to exist transferred later to plastic bags for temporary storage or to exist shipped they should first be allowed to remain completely immersed in preservative for at least 48 hours if formalin is used, or a calendar week if booze is used. The longer they are allowed to stay in preservative, the better. They should be loose and completely covered with plenty of liquid. Specimens which have been hardened in trays should as well exist allowed to soak in preservative for a day or 2 before being shipped or placed in plastic bags for storage. If space is no problem, preserved specimens are best kept in glass containers. Bond-top jars with a glass height and rubber gasket are best. Fruit jars with a metallic screwtop lid may be used only should be carefully watched for rust and evaporation. Drinking glass jars with polyethylene lids and liners are more normally used in collections, since the lids form a tight seal and are easier to obtain than the traditional bail-elevation jars. Metal containers should exist used simply for temporary storage unless coated on the inside with methane series, "Bakvar", or some other rustproof material.
Specimens should be loosely packed and completely covered with liquid. Containers must exist periodically checked for evaporation and refilled if necessary. At the get-go sign of decomposition the afflicted specimen should be removed and thrown away, or deep cuts made into the body cavity and placed alone in a large container with enough of fresh preservative. A green spot on the belly of a snake or cadger indicates a rotten gall float which should exist cut out. Whatsoever specimen that floats in the preservative contains air or other gases and is not properly preserved. It should be squeezed or slit to permit the gases to escape and the preservative to enter.
When traveling in the field, it is oft impossible to carry along a large number of drinking glass jars for storage. If specimens are well preserved and have been immersed in preservative for several days, it is safety to store them in plastic bags for a menses of several months. Plastic numberless are cheap and sold in diverse sizes. Whirl-Pak Bags are ideal for field collections, as they are fabricated of heavy-duty polyethylene and are leakproof when properly closed. Specimens should be wrapped loosely in cheesecloth. An piece of cake method is to cut a strip of cloth, lay it flat on a table and arrange the specimens in a row on the cloth with an inch or two betwixt them.
The strip should and then be rolled loosely with the specimens. Put the coil in a plastic bag and add plenty preservative to soak the cloth and accept a little free liquid in the bottom. Twist the open up end of the purse and wrap it tightly with a rubber band. Many museums use heat-sealed rolls of broad polytehylene tubing, which allows bags in any size with heat-sealed seams. This is an efficient mode to handle large numbers of specimens for shipping. A number of such bags may be stored in a metal tin, merely care should be taken to put bags with large, heavy specimens on the bottom.
Shipping: If your collection has been stored in plastic bags, simply fill up upward the container in which the numberless are stored with wads of textile or cotton so the bags will not knock well-nigh in transit, fasten the lid down tightly, and put the container in a wooden or heavy cardboard box for shipment. If plastic bags are not available, wrap the specimens loosely in cheesecloth and pack them carefully in a h2o-tight metal or plastic (polyethylene) container. If the bundles do not make full up the container, fill it up with wads of cloth or cotton. Never use paper, leaves or forest chips. Pour in enough preservative to soak the textile. No free liquid is necessary if the specimens are well preserved. Small bottles with tight screw caps containing tadpoles, salamander larvae or other delicate specimens should be wrapped with textile to foreclose breakage, and placed amidst the bundles of wrapped specimens.
Source: https://lsa.umich.edu/ummz/herps/collections/preservation-techniques.html
Posted by: weissfroned.blogspot.com

0 Response to "What Is Used For Preservation Of Dissection Animals Now"
Post a Comment